About Our Aesthetic Clinic
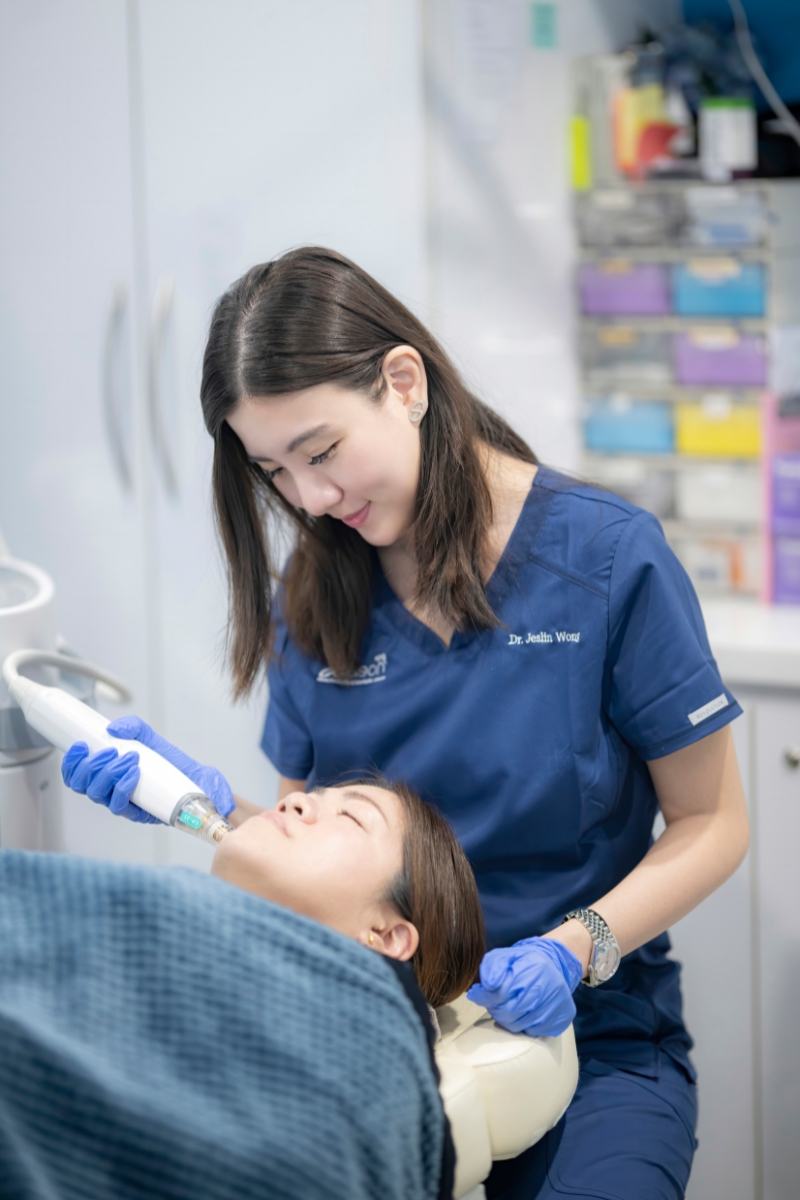
AEON Medical and Aesthetic Centre (AEON) is a fully medical licensed aesthetic clinic located in Orchard Road, Singapore. The clinic is helmed by Dr Jeslin Wong, with 7 years of medical and aesthetic practices under her belt. Dr Jeslin is always focused on providing medical treatments that are safe and effective. Patient's experiences pre and post treatment matters a lot to the doctor.
When at our clinic, all patients are assessed by Dr. Jeslin through a formal consultation session. We might also register your skin and facial images with our state-of-the-art VISIA (an advanced skin analysis system that provides a very in-depth and comprehensive analysis) to document your treatment progress over time.
All our treatment plans will be recommended based on your needs and concern. This may include a single or combination treatment of topical, oral, and non-invasive procedures.
Your Journey to a Better Skin
What’s Your Skin Concern?
Get the Latest News
Follow Us on Social Media
Get the Latest News
Follow Us on Social Media
Get the Latest News
Follow Us on Social Media
Frequently Asked Questions
It is important to choose the right skin aesthetic clinic in Singapore for your skin concerns. First, make sure that the medical team holds the right credentials and expertise needed for quality care. Then, find out if the clinic provides different services and adheres to local health regulations. AEON Medical & Aesthetic Centre is a fully licensed medical aesthetic clinic led by Dr Jeslin Wong, with 7 years of medical and aesthetic practices under her belt.
All patients are assessed by Dr. Jeslin through a formal consultation session, and the treatment plans will be recommended based on your needs and concerns.